Electron properties
Recreations of historic electron measurements, establishing the charge and mass of the electron.
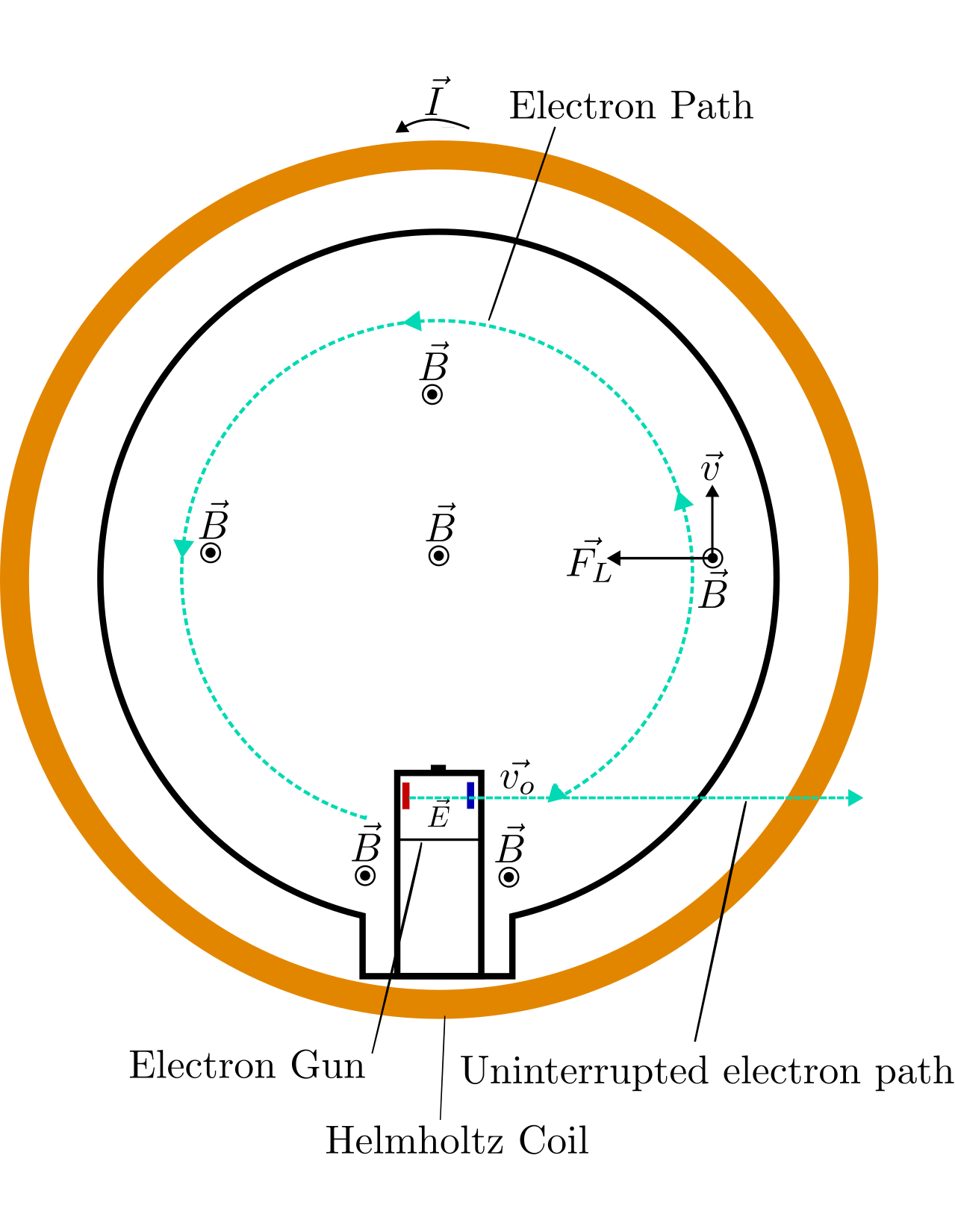
The charged family: verifying and quantizing electrons using electromagnetic forces
Author: Braedon Larsen
Abstract: When exposed to a magnetic field, charged particles such as electrons experience a force. If that particle is also moving at the time that force becomes a force known as a Lorentz force, which for the case of a magnetic field generated by a Helmholtz coil, will cause electrons to engage in cyclotron motion. Determining the radius of this motion and knowing the velocity and magnetic field strength can allow the charge to mass ratio to determined. As the average radius of motion and the square root of accelerating voltage over the magnetic field strength has a linear relationship that is proportional to the ratio of electron charge and mass. The determined ratio was found to be Ratio = (1.004 \(\pm\) .2509) \(\times\) 10\(^{11}\) C/kg .
When ionized oil proved quantization: data analysis techniques in ionized drops of oil for determining electron charge
Author: Braedon Larsen
Abstract: The charge on an electron is also referred to as the elementary charge e and is the quantum for the charge on a particle. Because of the quantization, charge on a particle will be an integer multiple of the elementary charge. By recording the velocities of ionized oil drops when naturally falling and when under the influence of a known electric field, then normalizing these charges, their existence as integer multiples of a single value can be seen. This value is the elementary charge and through the analysis done in this report was found to be \(e\) = (1.569 \(\pm\) .0913) \(\times\) 10\(^{-19}\) C